Recent
Recent
Top Stories
Top Stories
Recommended
Recommended
Read This Next
Read This Next
Learning Resources
Learning Resources
Sponsored Content
Featured Media
Featured Media
Home
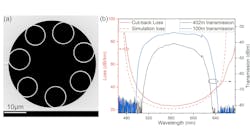
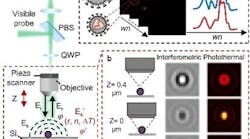
Photonics Hot List: April 19, 2024
April 19, 2024
Home
Photonics Hot List: April 12, 2024
April 12, 2024
Home
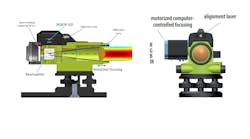
Photonics Hot List: April 5, 2024
April 5, 2024